

ISSN 1004-5759 CN 62-1105/S


草业学报 ›› 2026, Vol. 35 ›› Issue (5): 185-195.DOI: 10.11686/cyxb2025207
• 研究论文 • 上一篇
魏明( ), 吴欣蕊, 吴璇, 李昊, 伍国强, 张伟杰, 程子义
), 吴欣蕊, 吴璇, 李昊, 伍国强, 张伟杰, 程子义
收稿日期:2025-05-26
修回日期:2025-07-18
出版日期:2026-05-20
发布日期:2026-03-11
通讯作者:
魏明
作者简介:Corresponding author. E-mail: weim12@126.com基金资助:
Ming WEI( ), Xin-rui WU, Xuan WU, Hao LI, Guo-qiang WU, Wei-jie ZHANG, Zi-yi CHENG
), Xin-rui WU, Xuan WU, Hao LI, Guo-qiang WU, Wei-jie ZHANG, Zi-yi CHENG
Received:2025-05-26
Revised:2025-07-18
Online:2026-05-20
Published:2026-03-11
Contact:
Ming WEI
摘要:
甜菜作为我国北方盐碱地改良的先锋作物,其关键耐盐基因的挖掘对作物遗传改良具有重要理论价值。研究显示,甜菜碱醛脱氢酶基因家族BvBADHs通过生物合成甘氨酸甜菜碱可能在甜菜盐胁迫中发挥作用。本研究以耐盐甜菜品种‘甘糖7号’为材料,克隆该家族BvBADH2基因,利用农杆菌介导法构建过表达拟南芥株系,系统验证和解析其耐盐调控机制。基因克隆及生物信息学分析显示:BvBADH2 编码序列(CDS)全长1512 bp,编码503个氨基酸;预测蛋白相对分子量为54.78 kDa,等电点(pI)为5.37,不稳定指数为31.62;含有保守的乙醛脱氢酶结构域(Aldedh, PF00171),且启动子区域具有多种非生物胁迫响应的顺式作用元件。转基因拟南芥盐胁迫表型鉴定发现,150 mmol·L-1 NaCl胁迫下,过量表达株系OE1与OE3的根长、鲜重和叶绿素含量较野生型分别显著增加129.1%、125.0%、104.8%和137.5%、126.3%、107.2% (P<0.01)。进一步研究揭示,OE1/OE3通过特异性积累甘氨酸甜菜碱、脯氨酸和可溶性糖等渗透相容性物质,有效维持细胞渗透势稳态;同时动态激活超氧化物歧化酶、过氧化物酶和过氧化氢酶活性,显著抑制活性氧积累和缓解膜脂过氧化损伤。上述结果表明,BvBADH2通过“渗透-抗氧化协同调控网络”赋予植物耐盐性,首次在模式植物中验证其功能保守性,为作物耐盐分子设计育种提供了关键酶基因资源。
魏明, 吴欣蕊, 吴璇, 李昊, 伍国强, 张伟杰, 程子义. 甜菜碱醛脱氢酶基因BvBADH2的克隆及在植物盐胁迫中的作用[J]. 草业学报, 2026, 35(5): 185-195.
Ming WEI, Xin-rui WU, Xuan WU, Hao LI, Guo-qiang WU, Wei-jie ZHANG, Zi-yi CHENG. Cloning of the betaine aldehyde dehydrogenase family BvBADH2 gene and its role in plant salt tolerance[J]. Acta Prataculturae Sinica, 2026, 35(5): 185-195.
| 引物名称Name of primer | 序列Sequence (5′-3′) | 用途Purpose |
|---|---|---|
| BvBADH2 F1 | ATGGCGATCCCAATACCT | 基因克隆 Gene cloning |
| BvBADH2 R1 | CAGTTTTGAGGGAGACTTGTAC | |
| BvBADH2 F2 | gaggacacgctcgagATGGCGATCCCAATACCT | 载体构建 Vector construction |
| BvBADH2 R2 | tttgtaatccccgggCAGTTTTGAGGGAGACTTGTAC | |
| BvBADH2 qRT F | GTCCTGTTGTCAGCAAGGGA | 实时定量PCR Quantitative real time PCR |
| BvBADH2 qRT R | GCATGGACGTGGAGACATCA | |
| AtACTIN qRT F | AGATCCAGGACAAGGAAGGTATTC | |
| AtACTIN qRT R | CGCAGGACCAAGTGAAGAGTAG | |
| 35S Pro F | GACGCACAATCCCACTATCC | 转基因植株鉴定 Identification of the transgene plants |
| BvBADH2 R3 | CTCGTCATCGGAACTAAATG |
表1 本研究所用引物
Table 1 Primers used in this study
| 引物名称Name of primer | 序列Sequence (5′-3′) | 用途Purpose |
|---|---|---|
| BvBADH2 F1 | ATGGCGATCCCAATACCT | 基因克隆 Gene cloning |
| BvBADH2 R1 | CAGTTTTGAGGGAGACTTGTAC | |
| BvBADH2 F2 | gaggacacgctcgagATGGCGATCCCAATACCT | 载体构建 Vector construction |
| BvBADH2 R2 | tttgtaatccccgggCAGTTTTGAGGGAGACTTGTAC | |
| BvBADH2 qRT F | GTCCTGTTGTCAGCAAGGGA | 实时定量PCR Quantitative real time PCR |
| BvBADH2 qRT R | GCATGGACGTGGAGACATCA | |
| AtACTIN qRT F | AGATCCAGGACAAGGAAGGTATTC | |
| AtACTIN qRT R | CGCAGGACCAAGTGAAGAGTAG | |
| 35S Pro F | GACGCACAATCCCACTATCC | 转基因植株鉴定 Identification of the transgene plants |
| BvBADH2 R3 | CTCGTCATCGGAACTAAATG |

图1 BvBADH2分子克隆及编码蛋白序列分析A:BvBADH2基因扩增产物,大小1512 bp;B:BvBADH2全长编码序列分析。M:DL2000 DNA分子量标准;N:阴性对照;P:PCR产物;红色加粗氨基酸序列为乙醛脱氢酶保守结构域。下同。A: BvBADH2 gene amplification product, size 1512 bp. B: Analysis of the full-length coding sequence of BvBADH2. M: DNA marker DL2000; N: Negative control; P: PCR product. Red crude amino acid sequence is aldehyde dehydrogenases conserved domain. The same below.
Fig.1 Molecular cloning and protein sequence analysis of BvBADH2
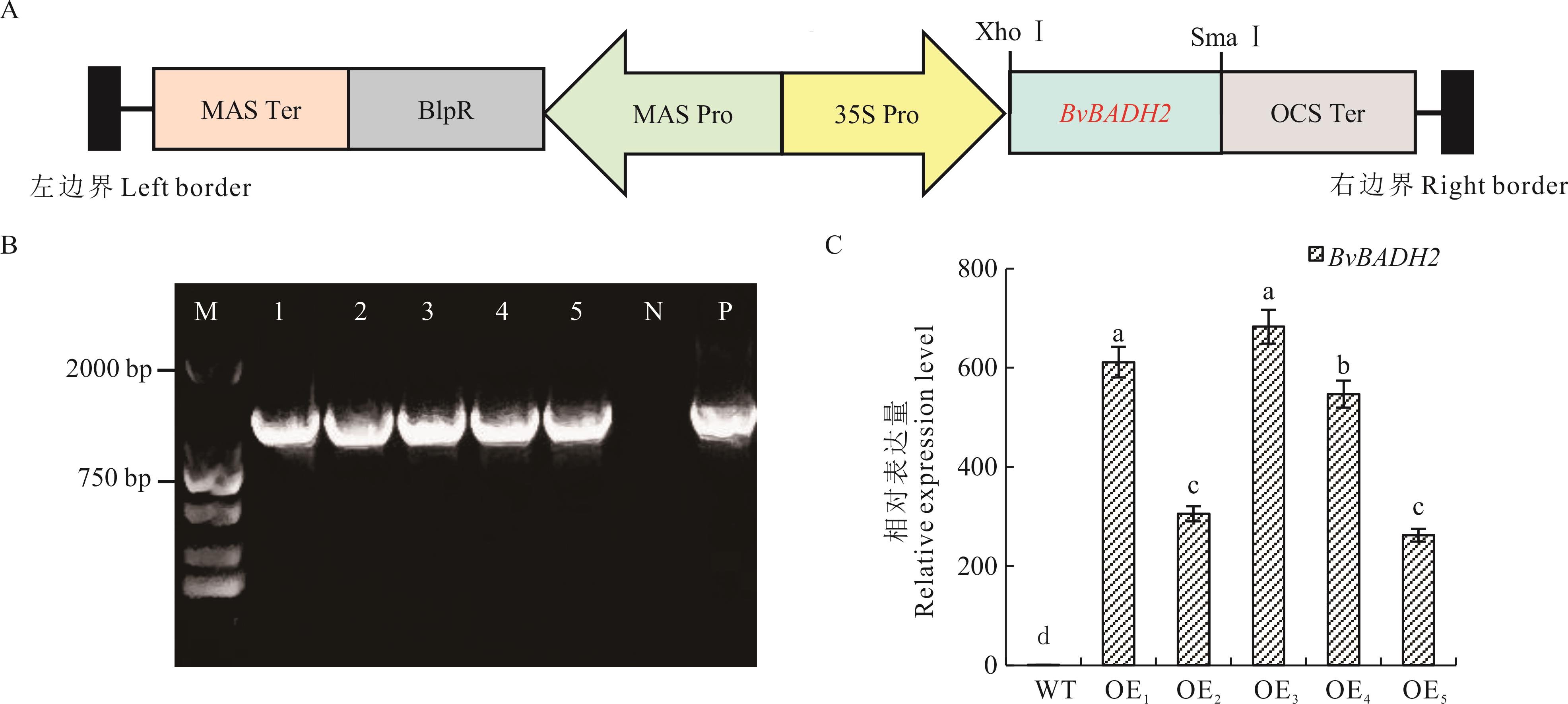
图2 BvBADH2过表达载体构建与转基因植株鉴定A: 双元载体T-DNA区域示意图Schematic diagram of the T-DNA region of the binary vector; MAS Ter: 甘露碱合成酶终止子Mannopine synthase terminator; BlpR: 类细菌素肽调控蛋白Bacteriocin-like peptide regulatory protein; MAS Pro: 甘露碱合成酶启动子Mannopine synthase promoter; 35S Pro: 花椰菜花叶病毒35S启动子Cauliflower mosaic virus 35S promoter; OCS Ter: 章鱼碱合成酶终止子Octopine synthase terminator. B: 转基因株系PCR鉴定PCR identification of transgenic lines; 1~5: 5个独立的过表达株系OE1~5 Five independent overexpression lines OE1-5;N: 阴性对照(野生型植株DNA) Negative control (wild-type plant DNA); P: 阳性对照(质粒DNA) Positive control (plasmid DNA); C: 转基因株系BvBADH2表达量的qRT-PCR检测qRT-PCR analysis of BvBADH2 relative expression levels in different transgenic lines; WT: 野生型Wild type. 不同小写字母代表不同株系间差异达到显著水平(P<0.05)。Different lowercase letters indicate significant differences among the lines (P<0.05).
Fig.2 Construction of the BvBADH2 overexpression vector and identification of transgenic plants
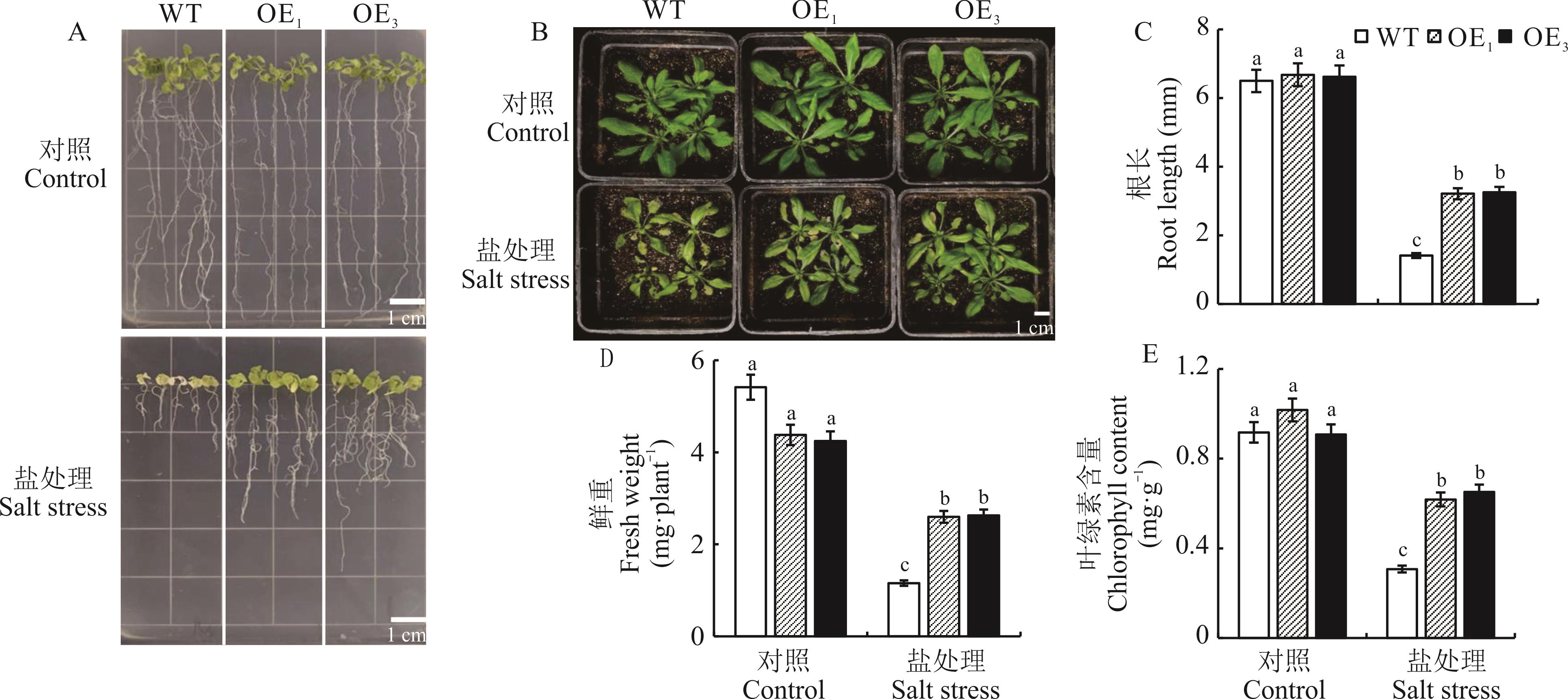
图3 过表达植株的盐胁迫表型分析不同小写字母表示对照与处理组不同株系间差异达到显著水平(P<0.05),下同。Different lowercase letters indicate significant differences among controls and treatments within different lines (P<0.05), the same below.
Fig.3 Phenotypic analysis of overexpression plants under salt stress
| [1] | Guang Z W, Gang N, Gu F, et al. Saline-alkali soil reclamation and utilization in China: progress and prospects. Frontiers of Agricultural Science and Engineering, 2024, 11(2): 216-228. |
| [2] | Zhang H M, Zhu J H, Gong Z Z, et al. Abiotic stress responses in plants. Nature Reviews Genetics, 2022, 23(2): 104-119. |
| [3] | Van Zelm E, Zhang Y, Testerink C. Salt tolerance mechanisms of plants. Annual Review of Plant Biology, 2020, 71(1): 403-433. |
| [4] | Munns R, Passioura J B, Colmer T D, et al. Osmotic adjustment and energy limitations to plant growth in saline soil. New Phytologist, 2020, 225(3): 1091-1096. |
| [5] | Yang Y Q, Guo Y. Elucidating the molecular mechanisms mediating plant salt-stress responses. New Phytologist, 2018, 217(2): 523-539. |
| [6] | Yu B, Chao D Y, Zhao Y. How plants sense and respond to osmotic stress. Journal of Integrative Plant Biology, 2024, 66(3): 394-423. |
| [7] | Chen T H, Murata N. Glycinebetaine: an effective protectant against abiotic stress in plants. Trends in Plant Science, 2008, 13(9): 499-505. |
| [8] | Fitzgerald T L, Waters D L, Henry R J. Betaine aldehyde dehydrogenase in plants. Plant Biology, 2009, 11(2): 119-130. |
| [9] | Chen T H, Murata N. Glycinebetaine protects plants against abiotic stress: mechanisms and biotechnological applications. Plant, Cell and Environment, 2011, 34(1): 1-20. |
| [10] | Xu Z J, Sun M L, Jiang X F, et al. Glycinebetaine biosynthesis in response to osmotic stress depends on jasmonate signaling in watermelon suspension cells. Frontiers in Plant Science, 2018, 9: 1469. |
| [11] | Weretilnyk E A, Hanson A D. Betaine aldehyde dehydrogenase from spinach leaves: purification, in vitro translation of the mRNA, and regulation by salinity. Archives of Biochemistry and Biophysics, 1989, 271(1): 56-63. |
| [12] | Mccue K F, Hanson A D. Salt-inducible betaine aldehyde dehydrogenase from sugar beet: cDNA cloning and expression. Plant Molecular Biology, 1992, 18(1): 1-11. |
| [13] | Ishitani M, Nakamura T, Han S Y, et al. Expression of the betaine aldehyde dehydrogenase gene in barley in response to osmotic stress and abscisic acid. Plant Molecular Biology, 1995, 27(2): 307-315. |
| [14] | Toshihide N, Sadaki Y, Yasunori M, et al. Expression of a betaine aldehyde dehydrogenase gene in rice, a glycinebetaine nonaccumulator, and possible localization of its protein in peroxisomes. The Plant Journal, 1997, 11(5): 1115-1120. |
| [15] | Li Q L, Gao X R, Yu X H, et al. Molecular cloning and characterization of betaine aldehyde dehydrogenase gene from Suaeda liaotungensis and its use in improved tolerance to salinity in transgenic tobacco. Biotechnology Letters, 2003, 25(17): 1431-1436. |
| [16] | Ali A, Ali Q, Iqbal M S, et al. Salt tolerance of potato genetically engineered with the Atriplex canescens BADH gene. Biologia Plantarum, 2020, 64: 271-279. |
| [17] | Yu Z J, Niu L, Cai Q A, et al. Improved salt-tolerance of transgenic soybean by stable over-expression of AhBADH gene from Atriplex hortensis. Plant Cell Reports, 2023, 42(8): 1291-1310. |
| [18] | Li H, Wu G Q, Wei M, et al. Genome-wide identification of the BvBADH gene family in sugar beet (Beta vulgaris) and their expression analysis under high salt stress. Biotechnology Bulletin, 2024, 40(2): 233-244. |
| 李昊, 伍国强, 魏明, 等. 甜菜BvBADH基因家族全基因组鉴定及其高盐胁迫下的表达分析. 生物技术通报, 2024, 40(2): 233-244. | |
| [19] | Han Y X, Wu G Q, Wei M, et al. The role of BADH in the response to abiotic stress in plants. Plant Physiology Journal, 2022, 58(2): 254-264. |
| 韩悦欣, 伍国强, 魏明, 等. BADH在植物响应非生物胁迫中的作用. 植物生理学报, 2022, 58(2): 254-264. | |
| [20] | Wu Z R, Zhang T Y, Li J N, et al. Genome-wide analysis of WD40 protein family and functional characterization of BvWD40-82 in sugar beet. Frontiers in Plant Science, 2023, 14: 1185440. |
| [21] | Bian C, Ji L Y, Xu W, et al. Research progress on bioactive substances of beets and their functions. Molecules, 2024, 29(19): 4756. |
| [22] | Wakeel A, Asif A R, Pitann B, et al. Proteome analysis of sugar beet (Beta vulgaris L.) elucidates constitutive adaptation during the first phase of salt stress. Journal of Plant Physiology, 2011, 168(6): 519-526. |
| [23] | Wang Y T, Liu H J, Wang M Q, et al. Salt tolerance in sugar beet: from impact analysis to adaptive mechanisms and future research. Plants, 2024, 13(21): 3018. |
| [24] | Höfgen R, Willmitzer L. Improved method for transformation of Agrobacterium tumefaciens with binary vector plasmids. Plant Molecular Biology Reporter, 1988, 6(2): 165-167. |
| [25] | Zhang X, Henriques R, Lin S S, et al. Agrobacterium-mediated transformation of Arabidopsis thaliana using the floral dip method. Nature Protocols, 2006, 1(2): 641-646. |
| [26] | Chomczynski P, Sacchi N.Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Analytical Biochemistry, 1987, 162(1): 156-159. |
| [27] | Ng H F, Ngeow Y F. A simple spreadsheet-based method for relative quantification using quantitative real-time PCR. Biochemistry and Molecular Biology Education, 2022, 50(1): 99-103. |
| [28] | Wellburn A R. The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. Journal of Plant Physiology, 1994, 144(3): 307-313. |
| [29] | Grieve C M, Grattan S R. Rapid assay for determination of water soluble quaternary ammonium compounds. Plant and Soil, 1983, 70(1): 303-307. |
| [30] | Bates L S, Waldren R P, Teare I D. Rapid determination of free proline for water-stress studies. Plant and Soil, 1973, 39(1): 205-207. |
| [31] | Dubois M, Gilles K A, Hamilton J K, et al. Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 1956, 28(3): 350-356. |
| [32] | Hodges D M, Delong J M, Forney C F, et al. Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta, 1999, 207(4): 604-611. |
| [33] | Gill S S, Tuteja N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiology and Biochemistry, 2010, 48(12): 909-930. |
| [34] | Chandran P R R, Sreedevi G S, Edwin B T, et al. Unveiling the key toxicity indicators and mechanisms on phytotoxicity of cerium dioxide nanoparticles in rice (Oryza sativa). Chemosphere, 2025, 376: 144270. |
| [35] | Thordal-Christensen H, Zhang Z G, Wei Y D, et al. Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley-powdery mildew interaction. The Plant Journal, 1997, 11(6): 1187-1194. |
| [36] | Thorsten J, Markus T, Christiane C, et al. Elicitor-stimulated ion fluxes and O2 - from the oxidative burst are essential components in triggering defense gene activation and phytoalexin synthesis in parsley. Proceedings of the National Academy of Sciences of the United States of America, 1997, 9(2): 207-220. |
| [37] | Schneider C A, Rasband W S, Eliceiri K W. NIH Image to ImageJ: 25 years of image analysis. Nature Methods, 2012, 9(7): 671-675. |
| [38] | Hasanuzzaman M, Fujita M. Plant responses and tolerance to salt stress: physiological and molecular interventions 2.0. International Journal of Molecular Sciences, 2023, 24(21): 15740. |
| [39] | Chen J H, Wang Y. Understanding the salinity resilience and productivity of halophytes in saline environments. Plant Science, 2024, 346: 112171. |
| [40] | Omari A F. Metabolic engineering of osmoprotectants to elucidate the mechanism(s) of salt stress tolerance in crop plants. Planta, 2021, 253(1): 24. |
| [41] | Ashraf M, Foolad M R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environmental and Experimental Botany, 2007, 59(2): 206-216. |
| [42] | Islam S, Mohammad F, Shakeel A, et al. Glycine betaine: a multifaceted protectant against salt stress in Indian mustard through ionic homeostasis, ROS scavenging and osmotic regulation. Physiologia Plantarum, 2024, 176(5): e14530. |
| [43] | Niazian M, Sadat-Noori S A, Tohidfar M, et al. Betaine aldehyde dehydrogenase (BADH) vs. flavodoxin (Fld): two important genes for enhancing plants stress tolerance and productivity. Frontiers in Plant Science, 2021, 12: 650215. |
| [44] | Mittler R. Oxidative stress, antioxidants and stress tolerance. Trends in Plant Science, 2002, 7(9): 405-410. |
| [45] | Fan W J, Zhang M, Zhang H X, et al. Improved tolerance to various abiotic stresses in transgenic sweet potato (Ipomoea batatas) expressing spinach betaine aldehyde dehydrogenase. PLoS One, 2012, 7(5): e37344. |
| [46] | Sun Y L, Liu X, Fu L S, et al. Overexpression of TaBADH increases salt tolerance in Arabidopsis. Canadian Journal of Plant Science, 2019, 99(4): 546-555. |
| [47] | Nathalie V, Christian H. Proline accumulation in plants: a review. Amino Acids, 2008, 35(4): 753-759. |
| [48] | De L T A, Montesinos-pereira D, Blasco B, et al. Influence of the proline metabolism and glycine betaine on tolerance to salt stress in tomato (Solanum lycopersicum L.) commercial genotypes. Journal of Plant Physiology, 2018, 231: 329-336. |
| [49] | Zhao P P, Zhao M, Gao X Y, et al. GhWRKY1bD improves drought tolerance by co-regulation of ABA, ROS, and proline homeostasis in cotton (Gossypium hirsutum). Industrial Crops and Products, 2024, 220: 14. |
| [50] | Miyashita Y, Good A G. NAD(H)-dependent glutamate dehydrogenase is essential for the survival of Arabidopsis thaliana during dark-induced carbon starvation. Journal of Experimental Botany, 2008, 59(3): 667-680. |
| [51] | Mittler R, Zandalinas S I, Fichman Y, et al. Reactive oxygen species signaling in plant stress responses. Nature Reviews Molecular Cell Biology, 2022, 23(10): 663-679. |
| [52] | Zhuang Y, Wei M, Ling C C, et al. EGY3 mediates chloroplastic ROS homeostasis and promotes retrograde signaling in response to salt stress in Arabidopsis. Cell Reports, 2021, 36(2): 109384. |
| [53] | Wang P, Liu W C, Han C, et al. Reactive oxygen species: multidimensional regulators of plant adaptation to abiotic stress and development. Journal of Integrative Plant Biology, 2024, 66(3): 330-367. |
| [54] | Kerchev P I, Van B F. Improving oxidative stress resilience in plants. The Plant Journal, 2022, 109(2): 359-372. |
| [1] | 马祥, 李中兴, 杨容尘, 琚泽亮, 贾志锋, 杨培志. 盐胁迫对不同耐盐性燕麦糖类及内源激素含量变化的影响[J]. 草业学报, 2026, 35(3): 235-244. |
| [2] | 杨寒星, 刘宁戈, 汤钰镂, 李欢, 朱一鸣, 郭家萌, 王浩, 邵瑞鑫, 王泳超, 杨青华. 水杨酸对高温、干旱及其复合胁迫下玉米抗氧化系统及光合能力的调控效应[J]. 草业学报, 2026, 35(1): 79-92. |
| [3] | 刘沂欣, 隋晓青, 王鑫尧, 郎梦卿, 孙凌子寅, 吉尔尔格. 外源褪黑素对盐胁迫下紫花苜蓿的缓解作用[J]. 草业学报, 2025, 34(9): 206-214. |
| [4] | 卢天一, 艾艳梅, 汪洋, 那萌, 徐尚起, 周际海. 镉污染土壤中水稻的镉富集特征和生长响应[J]. 草业学报, 2025, 34(8): 66-78. |
| [5] | 雍嘉仪, 马霜, 马风华, 赵小娜, 张译尹, 胡海英. 干旱及复水对河北木蓝生物量分配与渗透调节特征的影响[J]. 草业学报, 2025, 34(7): 158-170. |
| [6] | 张晴晴, 马兴羽, 鲁艳, 赵广兴, 曾凡江, 黄彩变. 沙化盐渍土地不同生长时期油莎豆的耐盐性差异研究[J]. 草业学报, 2025, 34(6): 168-180. |
| [7] | 王小风, 马步东, 黄海霞, 罗永忠, 齐建伟, 邓卓. 干旱胁迫及复水对裸果木幼苗生理特性的影响[J]. 草业学报, 2025, 34(4): 93-103. |
| [8] | 王梦琦, 王菲, 赵琬璐, 刘彦奇, 崔灿, 严俊鑫. 不同浓度硅、钙对留兰香幼苗生长和生理特性的影响[J]. 草业学报, 2025, 34(3): 154-163. |
| [9] | 田戈, 南丽丽, 王利群, 马香香, 何蓉, 郭佳雨. 盐胁迫下外源ABA对红豆草幼苗生长与生理特性的影响[J]. 草业学报, 2025, 34(10): 95-106. |
| [10] | 蔡文祺, 李淑霞, 王晓彤, 宋文学, 麻旭霞, 马小梅, 李小红, 代昕瑶. 外源褪黑素与乙烯交互对盐胁迫下紫花苜蓿幼苗生长和生理特性的影响[J]. 草业学报, 2025, 34(1): 80-93. |
| [11] | 张震欢, 姚立蓉, 汪军成, 司二静, 张宏, 杨轲, 马小乐, 孟亚雄, 王化俊, 李葆春. 盐生草AKR基因家族成员的鉴定及根系盐胁迫响应基因HgAKR42639的耐盐分析[J]. 草业学报, 2024, 33(7): 68-83. |
| [12] | 王萌, 鲁雪莉, 王菊英, 张梦超, 宋奕汝, 孟晨, 张莉, 徐宗昌. 小黑麦种质萌发期苗期耐盐资源评价与筛选[J]. 草业学报, 2024, 33(5): 58-68. |
| [13] | 张译尹, 李雪颖, 王斌, 宋珂辰, 兰剑, 胡海英. 盐胁迫对不同种质小黑麦幼苗水分利用效率和渗透调节的影响[J]. 草业学报, 2024, 33(4): 87-98. |
| [14] | 郭文婷, 王国华, 缑倩倩. 钠盐胁迫对藜科一年生草本植物种子萌发和幼苗生长的影响[J]. 草业学报, 2023, 32(3): 128-141. |
| [15] | 赵艳兰, 曾鑫奕, 弓晋超, 李香君, 李旭旭, 刘珊, 张新全, 周冀琼. 丛枝菌根真菌接种对白车轴草耐盐性的影响[J]. 草业学报, 2023, 32(3): 179-188. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||